
Preparing for the next pandemic: scientists discover a new class of influenza antivirals
Researchers from Leiden University, University of York, University of Barcelona/IQTCUB/ICREA and The Francis Crick Institute report a new class of experimental compounds that powerfully block influenza viruses. The findings offer a potential new strategy for treating flu infections and preparing for future pandemics.
The study, published in Proceedings of the National Academy of Sciences (PNAS), describes a family of molecules known as 'sugar aziridines.' These compounds are inspired by the widely used flu drug Oseltamivir (Tamiflu), but are designed to act in a fundamentally different way.
A stronger way to stop the flu virus
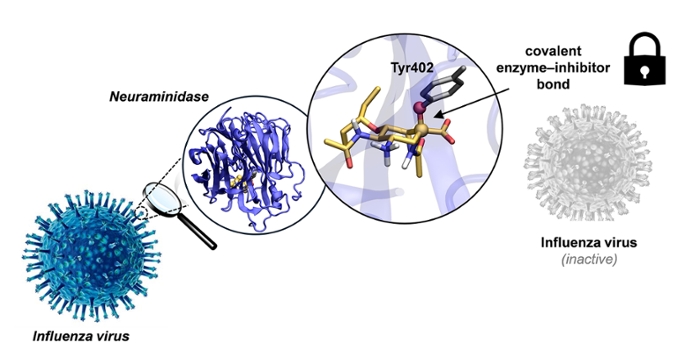
Influenza viruses rely on a surface enzyme called neuraminidase to spread from infected cells to healthy ones. Neuraminidase is also the target of today’s frontline flu medicines, which many countries stockpile as part of their pandemic preparedness plans. Current antiviral drugs, including oseltamivir, work by temporarily blocking this enzyme.
The newly developed sugar aziridines go a step further. First, they mimic a brief, critical stage in the enzyme’s chemical reaction, called the transition state, which allows the compounds to bind very tightly. Then, unlike existing drugs, they form a covalent (permanent) bond with the enzyme, effectively locking it in an inactive state.
Inspired by Tamiflu — redesigned to go further
To create this new class of inhibitors, the researchers modified the Tamiflu molecule by replacing a small chemical group (an alkene) with an aziridine ring in the appropriate configuration. This ring acts as a reactive “warhead,” enabling the permanent bond with the enzyme and thereby overcoming the reversible action of current flu drugs.
In lab tests following World Health Organization (WHO) guidelines, several of the compounds strongly blocked flu viruses from infecting cells. They were particularly effective against H3N2, one of the main viruses responsible for seasonal flu in humans, but also effectively inhibited neuraminidase from avian influenza strains such as H5N1, also known as bird flu.
Beyond treatment: improving vaccines and pandemic preparedness
In addition to their potential as flu treatments, sugar aziridines can also serve as versatile research tools. They allow scientists to label, image, and measure active neuraminidase in complex samples, including seasonal influenza vaccines.
Looking ahead, because the aziridine nitrogen can be modified, these molecules could form the basis for next-generation neuraminidase inhibitors and custom probes for research, diagnostics, and vaccine quality control. This could improve and accelerate the measurement of neuraminidase content and activity, key factors in how effectively vaccines protect against influenza.
Collaboration across boarders and disciplines
The project combined expertise in chemical synthesis, computational modelling, structural biology and virology, supported by European Union ERC-Synergy funding.
Professor Davies (University of York) comments: ‘For me, the beauty of this work is the synergy of the collaboration: synthesis, computation, three-dimensional structures and biology coming together to deliver a potent new class of anti-influenza agents.’
Professor Rovira (University of Barcelona) adds: ‘Our combined study let us watch these molecules “shut down” neuraminidase at the atomic level—first by fitting the enzyme’s transition state, then by enabling a covalent lock. It’s exciting to see computation help design inhibitors that can also be turned into imaging probes.’
From laboratory discovery to future flu medicines
The compounds are currently in the research phase and are not yet approved medicines. Further studies will be needed to evaluate safety, effectiveness in animals and humans, and long-term clinical potential.
Leiden Professor Overkleeft emphasises the next steps: ‘We are keen to develop the technology towards clinical application further. This will not be easy, as drug development is a lengthy and costly route where failure is more likely than success. Yet, the unique mode of action of our sugar aziridines, which we are in the process of patent-protecting, gives us a real edge over competing solutions.’
